AN OVERVIEW OF SOLID ELECTROLYTES FOR LITHIUM-ION BATTERIES BASED ON NASICON MATERIALS
Main Article Content
Abstract
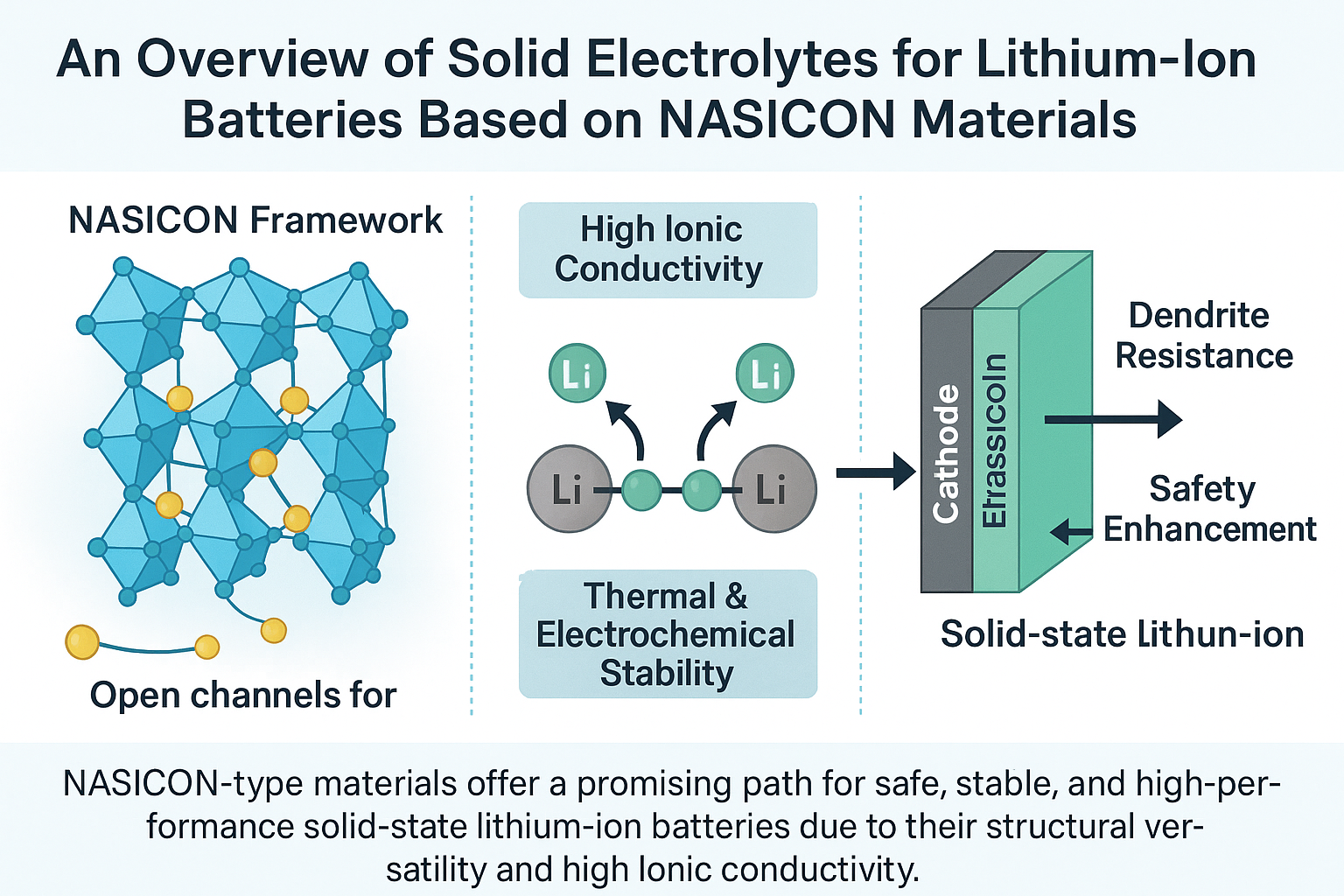
Lithium-ion solid electrolyte batteries have attracted great attention to replacing liquid electrolytes due to their cycling electrochemical properties, durable stability and safety. Numerous studies have been investigated to increase the performance of ionic-conductivity and enhance the stability of solid-state electrolytes, and its commercialization is broadly spread. This review article highlights the progress in developing NASICON-type solid electrolytes, which are made by solid-state reactions and wet-chemical procedures. Initially, researchers focused on the crystal structure, strength, and electrochemical properties of LiZr2(PO4)3, but recent studies have shifted towards investigating the electrical conductivity, crystal structure and its compatibility with Li metal. Also, the indigenous research on LiTi2(PO4)3 was pivoted on densification of the solid electrolyte to improve ionic conductivity by partial substitution of Ti4+ with Al3+ or B3+ now yields a promising ionic conductivity. LiGe2(PO4)3 is yet another Li-based NASICON electrolyte that, from the early stage, received extensive research majoring on the partial substitution of Al3+ with Ge4+ with the formula Li1+xAlxGe2-x(PO4)3 to stabilize the crystal structure following experimental and computational analysis. Improved ionic conductivity currently increasing as a result of improved Li intensity affected by aliovalent Al3+ substitution. Numerous research efforts have been explored to improve the ionic conductivity and elevate the electrochemical and thermal stabilities of NASICON-type electrolytes. It deems it fit to bring forth a cascade of promising results that so far have been recorded in both the early and the current research efforts in improving the suitability of NASICON-type electrolytes.
Downloads
Article Details
Section

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
How to Cite
References
[1] Z. Zhang, X. Wang, X. Li, J. Zhao, G. Liu, W. Yu, et al., “Review on composite solid electrolytes for solid-state lithium-ion batteries,” Mater. Today Sustain., vol. 21, p. 100316, Mar. 2023, doi: 10.1016/j.mtsust.2023.100316.
[2] M. Ma, M. Zhang, B. Jiang, Y. Du, B. Hu, and C. Sun, “A review of all-solid-state electrolytes for lithium batteries: high-voltage cathode materials, solid-state electrolytes and electrode-electrolyte interfaces,” Mater. Chem. Front., vol. 7, no. 7, pp. 1268–1297, 2023, doi: 10.1039/D2QM01071B.
[3] C. Li, R. Li, K. Liu, R. Si, Z. Zhang, and Y. Hu, “NaSICON: A promising solid electrolyte for solid‐state sodium batteries,” Interdiscip. Mater., vol. 1, no. 3, pp. 396–416, 2022, doi: 10.1002/idm2.12044.
[4] J. Jiang and J. Liu, “Iron anode‐based aqueous electrochemical energy storage devices: recent advances and future perspectives,” Interdiscip. Mater., vol. 1, no. 1, pp. 116–139, 2022, doi: 10.1002/idm2.12007.
[5] N. Boaretto, I. Garbayo, S. Valiyaveettil-SobhanRaj, A. Quintela, C. Li, M. Casas-Cabanas et al., “Lithium solid-state batteries: State-of-the-art and challenges for materials, interfaces and processing,” J. Power Sources, vol. 502, p. 229919, 2021, doi: 10.1016/j.jpowsour.2021.229919.
[6] C. Li, Z. Wang, Z. He, Y. Li, J. Mao, K. Dai, et al., “An advance review of solid-state battery: Challenges, progress and prospects,” Sustain. Mater. Technol., vol. 29, p. e00297, 2021, doi: 10.1016/j.susmat.2021.e00297.
[7] L. Wang, J. Li, G. Lu, W. Li, Q. Tao, C. Shi et al., “Fundamentals of electrolytes for solid-state batteries: challenges and perspectives,” Front. Mater., vol. 7, p. 111, 2020, doi: 10.3389/fmats.2020.00111.
[8] G. Yang, C. Abraham, Y. Ma, M. Lee, E. Helfrick, D. Oh, et al., “Advances in materials design for all-solid-state batteries: from bulk to thin films,” Appl. Sci., vol. 10, no. 14, p. 4727, 2020, doi: 10.3390/app10144727.
[9] H. Zhu, A. Prasad, S. Doja, L. Bichler, and J. Liu, “Spark plasma sintering of lithium aluminum germanium phosphate solid electrolyte and its electrochemical properties,” Nanomaterials, vol. 9, no. 8, p. 1086, 2019, doi: 10.3390/nano9081086.
[10] L. Zhang, Y. Liu, Y. You, A. Vinu, and L. Mai, “NASICONs‐type solid‐state electrolytes: The history, physicochemical properties, and challenges,” Interdiscip. Mater., vol. 2, no. 1, pp. 91–110, 2023, doi: 10.1002/idm2.12046.
[11] A. Jonderian and E. McCalla, “The role of metal substitutions in the development of Li batteries, part II: solid electrolytes,” Mater. Adv., vol. 2, no. 9, pp. 2846–2875, 2021, doi: 10.1039/D1MA00082A.
[12] J. A. Dias, S. H. Santagneli, and Y. Messaddeq, “Methods for lithium ion NASICON preparation: from solid-state synthesis to highly conductive glass-ceramics,” J. Phys. Chem. C, vol. 124, no. 49, pp. 26518–26539, 2020, doi: 10.1021/acs.jpcc.0c07385.
[13] A. Banik, T. Famprikis, M. Ghidiu, S. Ohno, M. A. Kraft, and W. G. Zeier, “On the underestimated influence of synthetic conditions in solid ionic conductors,” Chem. Sci., vol. 12, no. 18, pp. 6238–6263, 2021, doi: 10.1039/D0SC06553F.
[14] D. C. Gunduz, R. Schierholz, S. Yu, H. Tempel, H. Kungl, and R.-A. Eichel, “Combined quantitative microscopy on the microstructure and phase evolution in Li1.3Al 0.3Ti1.7(PO4)3 ceramics,” J. Adv. Ceram., vol. 9, pp. 149–161, 2020, doi: 10.1007/s40145-019-0354-0.
[15] A. Rossbach, F. Tietz, and S. Grieshammer, “Structural and transport properties of lithium-conducting NASICON materials,” J. Power Sources, vol. 391, pp. 1–9, 2018, doi: 10.1016/j.jpowsour.2018.04.059.
[16] F. Zheng, M. Kotobuki, S. Song, M. O. Lai, and L. Lu, “Review on solid electrolytes for all-solid-state lithium-ion batteries,” J. Power Sources, vol. 389, pp. 198–213, 2018, doi: 10.1016/j.jpowsour.2018.04.022.
[17] D. H. S. Tan, A. Banerjee, Z. Chen, and Y. S. Meng, “From nanoscale interface characterization to sustainable energy storage using all-solid-state batteries,” Nat. Nanotechnol., vol. 15, no. 3, pp. 170–180, 2020, doi: 10.1038/s41565-020-0657-x.
[18] S. He, Y. Xu, B. Zhang, X. Sun, Y. Chen, and Y. Jin, “Unique rhombus-like precursor for synthesis of Li1.3Al 0.3Ti1.7(PO4)3 solid electrolyte with high ionic conductivity,” Chem. Eng. J., vol. 345, pp. 483–491, 2018, doi: 10.1016/j.cej.2018.03.151.
[19] Y. Arinicheva, M. Wolff, S. Lobe, C. Dellen, D. Fattakhova-Rohlfing, O. Guillon et al., “Ceramics for electrochemical storage,” in Advanced ceramics for energy conversion and storage, Elsevier, 2020, pp. 549–709, doi: 10.1016/B978-0-08-102726-4.00010-7.
[20] L. Liu, D. Zhang, X. Xu, Z. Liu, and J. Liu, “Challenges and development of composite solid electrolytes for all-solid-state lithium batteries,” Chem. Res. Chinese Univ., vol. 37, pp. 210–231, 2021, doi: 10.1007/s40242-021-0007-z.
[21] M. Hou, F. Liang, K. Chen, Y. Dai, and D. Xue, “Challenges and perspectives of NASICON-type solid electrolytes for all-solid-state lithium batteries,” Nanotechnology, vol. 31, no. 13, p. 132003, 2020, doi: 10.1088/1361-6528/ab5be7.
[22] S. Sharma, V. Singh, R. K. Kotnala, and R. K. Dwivedi, “Comparative studies of pure BiFeO 3 prepared by sol–gel versus conventional solid-state-reaction method,” J. Mater. Sci. Mater. Electron., vol. 25, pp. 1915–1921, 2014, doi: 10.1007/s10854-014-1820-7.
[23] Y. B. Rao, K. K. Bharathi, and L. N. Patro, “Review on the synthesis and doping strategies in enhancing the Na ion conductivity of Na3Zr2Si2PO12 (NASICON) based solid electrolytes,” Solid State Ionics, vol. 366, p. 115671, 2021, doi: 10.1016/j.ssi.2021.115671.
[24] C. R. Mariappan, C. Galven, M.-P. Crosnier-Lopez, F. Le Berre, and O. Bohnke, “Synthesis of nanostructured LiTi2(PO4)3 powder by a Pechini-type polymerizable complex method,” J. Solid State Chem., vol. 179, no. 2, pp. 450–456, 2006, doi: 10.1016/j.jssc.2005.11.005.
[25] E. Zhao, F. Ma, Y. Jin, and K. Kanamura, “Pechini synthesis of high ionic conductivity Li1.3Al 0.3Ti1.7(PO4)3 solid electrolytes: The effect of dispersant,” J. Alloys Compd., vol. 680, pp. 646–653, 2016, doi: 10.1016/j.jallcom.2016.04.173.
[26] J. B. Goodenough, H.-P. Hong, and J. A. Kafalas, “Fast Na+-ion transport in skeleton structures,” Mater. Res. Bull., vol. 11, no. 2, pp. 203–220, 1976, doi: 10.1016/0025-5408(76)90077-5.
[27] N. a Anantharamulu, K. Koteswara Rao, G. Rambabu, B. Vijaya Kumar, V. Radha, and M. Vithal, “A wide-ranging review on Nasicon type materials,” J. Mater. Sci., vol. 46, pp. 2821–2837, 2011, doi: 10.1007/s10853-011-5302-5.
[28] M. I. Kimpa, M. Z. H. Mayzan, J. A. Yabagi, M. M. Nmaya, K. U. Isah, and M. A. Agam, “Review on material synthesis and characterization of sodium (Na) super-ionic conductor (NASICON),” in IOP Conference Series: Earth and Environmental Science, 2018, vol. 140, no. 1, p. 12156, doi: 10.1088/1755-1315/140/1/012156.
[29] H. Xie, J. B. Goodenough, and Y. Li, “Li1.2Zr1.9Ca0.1(PO4)3, a room-temperature Li-ion solid electrolyte,” J. Power Sources, vol. 196, no. 18, pp. 7760–7762, 2011, doi: 10.1016/j.jpowsour.2011.05.002.
[30] I. Hanghofer, B. Gadermaier, A. Wilkening, D. Rettenwander, and H. M. R. Wilkening, “Lithium ion dynamics in LiZr2(PO4)3 and Li1.4Ca0.2Zr1.8(PO4)3,” Dalt. Trans., vol. 48, no. 25, pp. 9376–9387, 2019, doi: 10.1039/C9DT01786K.
[31] Y. Li, M. Liu, K. Liu, and C. A. Wang, “High Li+ conduction in NASICON-type Li1+ xYxZr2− x (PO4)3 at room temperature,” J. Power Sources, vol. 240, pp. 50–53, 2013, doi: 10.1016/j.jpowsour.2013.03.175.
[32] Y. Li, W. Zhou, X. Chen, X. Lü, Z. Cui, S. Xin, et al., “Mastering the interface for advanced all-solid-state lithium rechargeable batteries,” Proc. Natl. Acad. Sci., vol. 113, no. 47, pp. 13313–13317, 2016, doi: 10.1073/pnas.161591211.
[33] Q. Zhou, B. Xu, P. H. Chien, Y. Li, B. Huang, N. Wu, et al., “NASICON Li1.2Mg0.1Zr1.9(PO4)3 solid electrolyte for an all‐solid‐state Li‐metal battery,” Small Methods, vol. 4, no. 12, p. 2000764, 2020, doi: 10.1002/smtd.202000764.
[34] Y. Noda, K. Nakano, H. Takeda, M. Kotobuki, L. Lu, and M. Nakayama, “Computational and experimental investigation of the electrochemical stability and Li-ion conduction mechanism of LiZr2(PO4)3,” Chem. Mater., vol. 29, no. 21, pp. 8983–8991, 2017, doi: 10.1021/acs.chemmater.7b01703.
[35] H. AONO, “Ionic conductivity and sinterability of lithium titanium phosphate system,” Solid State Ionics, vol. 40–41, pp. 38–42, Aug. 1990, doi: 10.1016/0167-2738(90)90282-V.
[36] E. Kazakevicius, T. Šalkus, A. Dindune, Z. Kanepe, J. Ronis, A. Kežionis, et al., “La-doped LiTi2(PO4)3 ceramics,” Solid State Ionics, Feb. 2008, doi: 10.1016/j.ssi.2007.12.025.
[37] J. Fu, “Superionic conductivity of glass-ceramics in the system Li2O-Al2O3-TiO2-P2O5,” Solid State Ionics, vol. 96, no. 3–4, pp. 195–200, Apr. 1997, doi: 10.1016/S0167-2738(97)00018-0.
[38] J. Fu, “Fast Li+ Ion Conduction in Li2O‐Al2O3‐TiO2‐SiO2‐P2O2 Glass‐Ceramics,” J. Am. Ceram. Soc., vol. 80, no. 7, pp. 1901–1903, Jul. 1997, doi: 10.1111/j.1151-2916.1997.tb03070.x.
[39] Y. Shimonishi, T. Zhang, N. Imanishi, D. Im, D. J. Lee, A. Hirano, et al., “A study on lithium/air secondary batteries—Stability of the NASICON-type lithium ion conducting solid electrolyte in alkaline aqueous solutions,” J. Power Sources, vol. 196, no. 11, pp. 5128–5132, Jun. 2011, doi: 10.1016/j.jpowsour.2011.02.023.
[40] Y. Shimonishi, T. Zhang, P. Johnson, N. Imanishi, A. Hirano, Y. Takeda, et al., “A study on lithium/air secondary batteries—Stability of NASICON-type glass ceramics in acid solutions,” J. Power Sources, vol. 195, no. 18, pp. 6187–6191, Sep. 2010, doi: 10.1016/j.jpowsour.2009.11.023.
[41] G. Tan, F. Wu, L. Li, Y. Liu, and R. Chen, “Magnetron Sputtering Preparation of Nitrogen-Incorporated Lithium–Aluminum–Titanium Phosphate Based Thin Film Electrolytes for All-Solid-State Lithium Ion Batteries,” J. Phys. Chem. C, vol. 116, no. 5, pp. 3817–3826, Feb. 2012, doi: 10.1021/jp207120s.
[42] H. Peng, H. Xie, and J. B. Goodenough, “Use of B2O3 to improve Li+-ion transport in LiTi2(PO4)3-based ceramics,” J. Power Sources, vol. 197, pp. 310–313, Jan. 2012, doi: 10.1016/j.jpowsour.2011.09.046.
[43] J. S. Thokchom and B. Kumar, “The effects of crystallization parameters on the ionic conductivity of a lithium aluminum germanium phosphate glass–ceramic,” J. Power Sources, vol. 195, no. 9, pp. 2870–2876, May 2010, doi: 10.1016/j.jpowsour.2009.11.037.
[44] X. Xu, Z. Wen, X. Wu, X. Yang, and Z. Gu, “Lithium Ion‐Conducting Glass–Ceramics of Li1.5Al0.5Ge1.5(PO4)3–x Li2O ( x =0.0–0.20) with Good Electrical and Electrochemical Properties,” J. Am. Ceram. Soc., vol. 90, no. 9, pp. 2802–2806, Sep. 2007, doi: 10.1111/j.1551-2916.2007.01827.x.
[45] A. Paolella, W. Zhu, G. Bertoni, A. Perea, H. Demers, S. Savoie, et al., “Toward an All‐Ceramic Cathode–Electrolyte Interface with Low‐Temperature Pressed NASICON Li1.5Al 0.5Ge1.5(PO4)3 Electrolyte,” Adv. Mater. Interfaces, vol. 7, no. 12, Jun. 2020, doi: 10.1002/admi.202000164.
[46] P. Hartmann, T. Leichtweiss, M. R. Busche, M. Schneider, M. Reich, J. Sann, et al., “Degradation of NASICON-Type Materials in Contact with Lithium Metal: Formation of Mixed Conducting Interphases (MCI) on Solid Electrolytes,” J. Phys. Chem. C, vol. 117, no. 41, pp. 21064–21074, Oct. 2013, doi: 10.1021/jp4051275.
[47] Y.-Y. Sun, Q. Zhang, L. Yan, T.-B. Wang, and P.-Y. Hou, “A review of interfaces within solid-state electrolytes: fundamentals, issues and advancements,” Chem. Eng. J., vol. 437, p. 135179, Jun. 2022, doi: 10.1016/j.cej.2022.135179.
[48] L. Xu, J. Li, W. Deng, H. Shuai, S. Li, Z. Xu, et al., “Garnet Solid Electrolyte for Advanced All‐Solid‐State Li Batteries,” Adv. Energy Mater., vol. 11, no. 2, Jan. 2021, doi: 10.1002/aenm.202000648.
[49] K. Subramanian, G. V. Alexander, K. Karthik, S. Patra, M. S. Indu, O. V. Sreejith, et al., “A brief review of recent advances in garnet structured solid electrolyte based lithium metal batteries,” J. Energy Storage, vol. 33, p. 102157, Jan. 2021, doi: 10.1016/j.est.2020.102157.
[50] M. I. Kimpa, M. Z. H. Bin Mayzan, F. Esa, J. A. Yabagi, M. M. Nmaya, and M. A. Bin Agam, “Physical characterization and electrical conductivity of Li1.2Ti1.8Al0.2(PO4)3 and Li1.2Ta0.9Al1.1(PO4)3 NASICON material,” Int. J. Integr. Eng., vol. 10, no. 9, Dec. 2018, doi: 10.30880/ijie.2018.10.09.020.
[51] T. Zangina, J. Hassan, K. A. Matori, R. S. Azis, C. E. Ndikilar, and F. H. Naning, “Dielectric Relaxation Analysis of Chemical Solid Electrolyte Lithium Aluminum Titanium Phosphate,” Asian J. Appl. Sci., vol. 11, no. 1, pp. 46–55, Dec. 2017, doi: 10.3923/ajaps.2018.46.55.
[52] M. Niazmand, Z. Khakpour, and A. Mortazavi, “Electrochemical properties of nanostructure NASICON synthesized by chemical routes: A comparison between coprecipitation and sol-gel,” J. Alloys Compd., vol. 798, pp. 311–319, Aug. 2019, doi: 10.1016/j.jallcom.2019.05.170.
[53] K. G. Schell, E. C. Bucharsky, F. Lemke, and M. J. Hoffmann, “Effect of calcination conditions on lithium conductivity in Li1.3Ti1.7Al0.3(PO4)3 prepared by sol-gel route,” Ionics (Kiel)., vol. 23, no. 4, pp. 821–827, Apr. 2017, doi: 10.1007/s11581-016-1883-y.
[54] P. Bhanja, C. Senthil, A. K. Patra, M. Sasidharan, and A. Bhaumik, “NASICON type ordered mesoporous lithium-aluminum-titanium-phosphate as electrode materials for lithium-ion batteries,” Microporous Mesoporous Mater., vol. 240, pp. 57–64, Mar. 2017, doi: 10.1016/j.micromeso.2016.11.005.
[55] S. Duluard, A. Paillassa, L. Puech, P. Vinatier, V. Turq, P. Rozier, et al., “Lithium conducting solid electrolyte Li1.3Al0.3Ti1.7(PO4)3 obtained via solution chemistry,” J. Eur. Ceram. Soc., vol. 33, no. 6, pp. 1145–1153, Jun. 2013, doi: 10.1016/j.jeurceramsoc.2012.08.005.
[56] L. Huang, Z. Wen, M. Wu, X. Wu, Y. Liu, and X. Wang, “Electrochemical properties of Li1.4Al0.4Ti1.6(PO4)3 synthesized by a co-precipitation method,” J. Power Sources, vol. 196, no. 16, pp. 6943–6946, Aug. 2011, doi: 10.1016/j.jpowsour.2010.11.140.
[57] Y. Zhu, T. Wu, J. Sun, and M. Kotobuki, “Highly conductive lithium aluminum germanium phosphate solid electrolyte prepared by sol-gel method and hot-pressing,” Solid State Ionics, vol. 350, p. 115320, Jul. 2020, doi: 10.1016/j.ssi.2020.115320.
[58] C. R. Mariappan, P. Kumar, A. Kumar, S. Indris, H. Ehrenberg, G. V. Prakash, et al., “Ionic conduction and dielectric properties of yttrium doped LiZr2(PO4)3 obtained by a Pechini-type polymerizable complex route,” Ceram. Int., vol. 44, no. 13, pp. 15509–15516, Sep. 2018, doi: 10.1016/j.ceramint.2018.05.211.
[59] G. B. Kunshina, I. V. Bocharova, and V. I. Ivanenko, “Effect of thermal treatment modes on ion-conducting properties of lithium-aluminum titanophosphate,” Russ. J. Appl. Chem., vol. 90, no. 3, pp. 374–379, Mar. 2017, doi: 10.1134/S1070427217030089.
[60] H. Zhu and J. Liu, “Emerging applications of spark plasma sintering in all solid-state lithium-ion batteries and beyond,” J. Power Sources, vol. 391, pp. 10–25, Jul. 2018, doi: 10.1016/j.jpowsour.2018.04.054.
[61] L. Hallopeau, D. Bregiroux, G. Rousse, D. Portehault, P. Stevens, G. Toussaint, et al., “Microwave-assisted reactive sintering and lithium ion conductivity of Li1.3Al0.3Ti1.7(PO4)3 solid electrolyte,” J. Power Sources, vol. 378, pp. 48–52, Feb. 2018, doi: 10.1016/j.jpowsour.2017.12.021.
[62] X. Wei, J. Rechtin, and E. Olevsky, “The Fabrication of All-Solid-State Lithium-Ion Batteries via Spark Plasma Sintering,” Metals (Basel)., vol. 7, no. 9, p. 372, Sep. 2017, doi: 10.3390/met7090372.
[63] J. Feng, H. Xia, M. O. Lai, and L. Lu, “NASICON-Structured LiGe2(PO4)3 with Improved Cyclability for High-Performance Lithium Batteries,” J. Phys. Chem. C, vol. 113, no. 47, pp. 20514–20520, Nov. 2009, doi: 10.1021/jp9085602.
[64] H. Aono, E. Sugimoto, Y. Sadaoka, N. Imanaka, and G. Adachi, “Electrical properties and sinterability for lithium germanium phosphate Li1+xMxGe2-x(PO4)3, M=Al, Cr, Ga, Fe, Sc, and in systems,” Bull. Chem. Soc. Jpn., vol. 65, no. 8, pp. 2200–2204, 1992.
[65] Y. Liu, J. Chen, and J. Gao, “Preparation and chemical compatibility of lithium aluminum germanium phosphate solid electrolyte,” Solid State Ionics, vol. 318, pp. 27–34, May 2018, doi: 10.1016/j.ssi.2017.10.016.
[66] M. Sugantha and U. V Varadaraju, “Ionic conductivity of Li+ ion conductors Li2M3+ M4+ P3O12,” Solid State Ionics, vol. 95, no. 3–4, pp. 201–205, 1997, doi: 10.1016/S0167-2738(96)00565-6.
[67] A. Venkateswara Rao, V. Veeraiah, A. V. Prasada Rao, and B. Kishore Babu, “Effect of Fe3+ doping on the structure and conductivity of LiTi2(PO4)3,” Res. Chem. Intermed., vol. 41, no. 4, pp. 2307–2315, Apr. 2015, doi: 10.1007/s11164-013-1348-0.
[68] R. Kahlaoui, K. Arbi, R. Jimenez, I. Sobrados, J. Sanz, and R. Ternane, “Influence of preparation temperature on ionic conductivity of titanium-defective Li1+4xTi2−x(PO4)3 NASICON-type materials,” J. Mater. Sci., vol. 55, no. 20, pp. 8464–8476, Jul. 2020, doi: 10.1007/s10853-020-04463-3.
[69] A. Belous, G. Kolbasov, L. Kovalenko, E. Boldyrev, S. Kobylianska, and B. Liniova, “All solid-state battery based on ceramic oxide electrolytes with perovskite and NASICON structure,” J. Solid State Electrochem., vol. 22, no. 8, pp. 2315–2320, Aug. 2018, doi: 10.1007/s10008-018-3943-x.
[70] A. Venkateswara Rao, V. Veeraiah, A. V. Prasada Rao, B. Kishore Babu, and K. V. Kumar, “Influence of Zr4+ doping on structural, spectroscopic and conductivity studies of lithium titanium phosphate,” Ceram. Int., vol. 40, no. 9, pp. 13911–13916, Nov. 2014, doi: 10.1016/j.ceramint.2014.05.111.
[71] Z. Li and X. Zhao, “Influence of excess lithium and sintering on the conductivity of Li1.3Al0.3Ti1.7(PO4)3,” Funct. Mater. Lett., vol. 12, no. 04, p. 1950047, Aug. 2019, doi: 10.1142/S1793604719500474.
[72] R. Kahlaoui, K. Arbi, R. Jimenez, I. Sobrados, J. Sanz, and R. Ternane, “Distribution and mobility of lithium in NASICON-type Li1-xTi2-xNbx(PO4)3 (0 ≤ x ≤ 0.5) compounds,” Mater. Res. Bull., vol. 101, pp. 146–154, May 2018, doi: 10.1016/j.materresbull.2018.01.022.
[73] H. Aono, E. Sugimoto, Y. Sadaoka, N. Imanaka, and G. Adachi, “Electrical properties and crystal structure of solid electrolyte based on lithium hafnium phosphate LiHf2(PO4)3,” Solid State Ionics, vol. 62, no. 3–4, pp. 309–316, Aug. 1993, doi: 10.1016/0167-2738(93)90387-I.
[74] R. Kahlaoui, K. Arbi, I. Sobrados, R. Jimenez, J. Sanz, and R. Ternane, “Cation Miscibility and Lithium Mobility in NASICON Li1+ xTi2– xScx(PO4)3 (0 ≤ x ≤ 0.5) Series: A Combined NMR and Impedance Study,” Inorg. Chem., vol. 56, no. 3, pp. 1216–1224, Feb. 2017, doi: 10.1021/acs.inorgchem.6b02274.
[75] A. Cassel, B. Fleutot, M. Courty, V. Viallet, and M. Morcrette, “Sol-gel synthesis and electrochemical properties extracted by phase inflection detection method of NASICON-type solid electrolytes LiZr2(PO4)3 and Li1.2Zr1.9Ca0.1(PO4)3,” Solid State Ionics, vol. 309, pp. 63–70, Oct. 2017, doi: 10.1016/j.ssi.2017.07.009.
[76] Y. Zhang, K. Chen, Y. Shen, Y. Lin, and C.-W. Nan, “Enhanced lithium-ion conductivity in a LiZr2(PO4)3 solid electrolyte by Al doping,” Ceram. Int., vol. 43, pp. S598–S602, Aug. 2017, doi: 10.1016/j.ceramint.2017.05.198.
[77] B. E. Francisco, C. R. Stoldt, and J.-C. M’Peko, “Lithium-Ion Trapping from Local Structural Distortions in Sodium Super Ionic Conductor (NASICON) Electrolytes,” Chem. Mater., vol. 26, no. 16, pp. 4741–4749, Aug. 2014, doi: 10.1021/cm5013872.
[78] E. Yi, K. Yoon, H.-A. Jung, T. Nakayama, M. Ji, and H. Hwang, “Fabrication and electrochemical properties of Li1.3Al0.3Ti1.7(PO4)3 solid electrolytes by sol-gel method,” Appl. Surf. Sci., vol. 473, pp. 622–626, Apr. 2019, doi: 10.1016/j.apsusc.2018.12.202.
[79] X. Xu, Z. Wen, X. Yang, and L. Chen, “Dense nanostructured solid electrolyte with high Li-ion conductivity by spark plasma sintering technique,” Mater. Res. Bull., vol. 43, no. 8–9, pp. 2334–2341, Aug. 2008, doi: 10.1016/j.materresbull.2007.08.007.
[80] S. Breuer, D. Prutsch, Q. Ma, V. Epp, F. Preishuber-Pflügl, F. Tietz, et al., “Separating bulk from grain boundary Li ion conductivity in the sol–gel prepared solid electrolyte Li1.5Al0.5Ti1.5(PO4)3,” J. Mater. Chem. A, vol. 3, no. 42, pp. 21343–21350, 2015, doi: 10.1039/C5TA06379E.
[81] P. Zhang, M. Matsui, A. Hirano, Y. Takeda, O. Yamamoto, and N. Imanishi, “Water-stable lithium ion conducting solid electrolyte of the Li1.4Al0.4Ti1.6−xGex(PO4)3 system (x=0–1.0) with NASICON-type structure,” Solid State Ionics, vol. 253, pp. 175–180, Dec. 2013, doi: 10.1016/j.ssi.2013.09.022.
[82] V. Ramar, S. Kumar, S. R. Sivakkumar, and P. Balaya, “NASICON-type La3+substituted LiZr2(PO4)3 with improved ionic conductivity as solid electrolyte,” Electrochim. Acta, vol. 271, pp. 120–126, May 2018, doi: 10.1016/j.electacta.2018.03.115.
[83] M. I. Kimpa, M. Z. H. Mayzan, F. Esa, J. A. Yabagi, M. M. Nmaya, and M. A. Agam, “Sol-Gel Synthesis and Electrical Characterization of Li1+ xAlxTi2-x(PO4)3 Solid Electrolytes,” J. Sci. Technol., vol. 9, no. 3, 2017.
[84] M. Perez-Estébanez, M. Peiteado, A. C. Caballero, F. J. Palomares, M. Nygren, and J. Isasi-Marín, “SPS driven lithium differential diffusion in NASICON-like structures,” Boletín la Soc. Española Cerámica y Vidr., vol. 55, no. 1, pp. 38–44, Jan. 2016, doi: 10.1016/j.bsecv.2015.11.005.
[85] C. Chang, Y. Il Lee, S. Hong, and H. Park, “Spark Plasma Sintering of LiTi2(PO4)3 ‐Based Solid Electrolytes,” J. Am. Ceram. Soc., vol. 88, no. 7, pp. 1803–1807, Jul. 2005, doi: 10.1111/j.1551-2916.2005.00246.x.
[86] Y. Nikodimos, M. C. Tsai, L. H. Abrha, H. H. Weldeyohannis, S. F. Chiu, H. K. Bezabh, et al., “Al–Sc dual-doped LiGe2(PO4)3 – a NASICON-type solid electrolyte with improved ionic conductivity,” J. Mater. Chem. A, vol. 8, no. 22, pp. 11302–11313, 2020, doi: 10.1039/D0TA00517G.
[87] H. R. Arjmandi and S. Grieshammer, “Defect formation and migration in Nasicon Li1+xAlxTi2−x(PO4)3,” Phys.